The ability of matter, to transmit light waves, is specified as transparency. If “something positioned behind something” can be relatively clearly recognizable, normally the material is considered transparent or lucent like in the case of window glass. This is the case for most gaseous, as well as many liquids substances and some solid materials. If the degree of transparency is dependent on the wavelength of the light, then the transparent substance is tinted through absorptive acting particles. For example, with certain metal oxide molecules in glass or coloured smoke. The higher the concentration of these particles, the less transparent the material becomes.
Translucence and opacity
Unlike transparent materials, are materials which transmit light, but like in the case with frosted glass where no objects can be recognizable behind it, considered translucent or transparent. It is considered opacity with non-translucent materials, like metal and wood. The opacity is a measurement for the obfuscation of materials and is the reciprocal of translucency.
Opalescence
Depending on the size of the scattering particles, a further so called opalescence can occur: the colourfulness of certain materials, which occurs through scattering and possibly interference of light on small structures in a material. Are the scattering particles smaller than the wave length, a strongly wave-dependent Rayleigh scattering occurs, where the scattering intensity substantially increases with a decreasing wave length. Therefore, a blue light is more strongly scattered than a red one, which for example causes the blue colour of the sky. When particles become larger than the wave length of the light, the Mie-scattering occurs instead where the wave length dependency of the scattering light intensity is less distinct. The colourfulness of the scattered white light is then lost. A passage from opalescence to opacity is observed.
The measurement
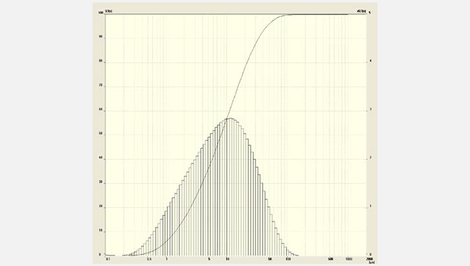
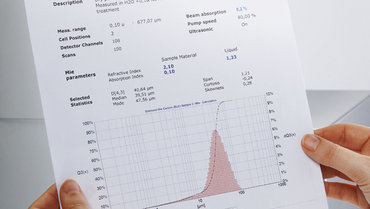
The example measurement illustrates a particle size distribution of ground cryolite which for example is used for the tinting of glass (frosted glass). Different visual impressions occur depending on the particle size.
The wet measuring was performed with a saturated cryolite solution, in which a background measurement with the ANALYSETTE 22 was carried out prior to adding the sample. Thereby a de-stabilizing of the solution was avoided which would have been observed with the same sample in pure water. Unstable solutions lead to a shifting of the measuring curve during successive measurements to smaller particle diameters. As an alternative, the sample can be measured in alcohol by using a small volume dispersion unit.
-
Download the FRITSCH-report as PDF file
Optical characteristics of transparent and opaque materials – influence of the particle size