The majority of all drugs have to - in order to unfold their full effectiveness - reach the bloodstream. As a rule, this happens orally, via the bowels, by injections or with infusions and with the so called new “pharmaceutical dosage forms”.
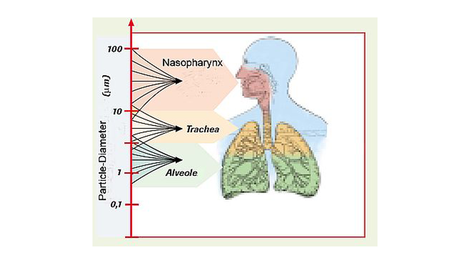
Considered are for example the transdermal systems which through the skin, a specific dose of medication is absorbed, or micronized powders; whereas the specific dosage is either by the lung (pulmonary) or with the mucous membrane of the nasal cavity (nasally) inhaled and distributed via the bloodstream.
The different bioavailability for the various pharmaceutical forms is herewith among other things clearly influenced by the particle size of the carrier substance, or the active ingredient itself, which is critical in regards to a sensible dosage recommendation.
In the meantime play powders as an independent type dosage form only a minor part, as a base material for other types of dosage forms they are still important though. Besides their significance for the effectiveness of drugs, the particle size distribution plays an important part in the production process (quality control) and in the development of suitable manufacturing methods and plants.
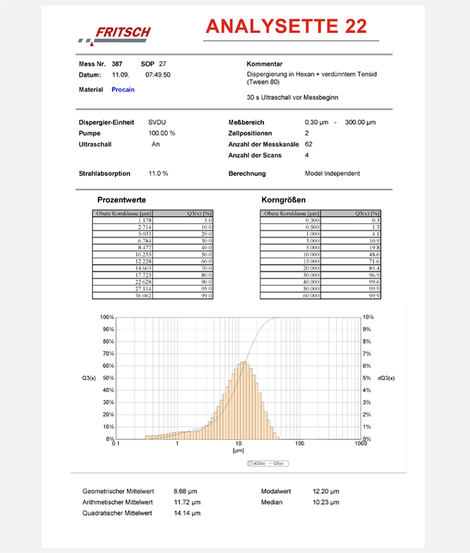
The example below for the particle size distribution of ampicillin, a semi-synthetic antibiotic from the group of beta-lactam antibiotics (penicillin), was respectively measured with a small volume dispersion unit in hexane, since the sample material dissolves in water and even other solvents. Prior to the actual measuring was an externally prepared suspension for 30 seconds pre-dispersed in an ultrasonic bath.
-
Download the FRITSCH-report as PDF file